 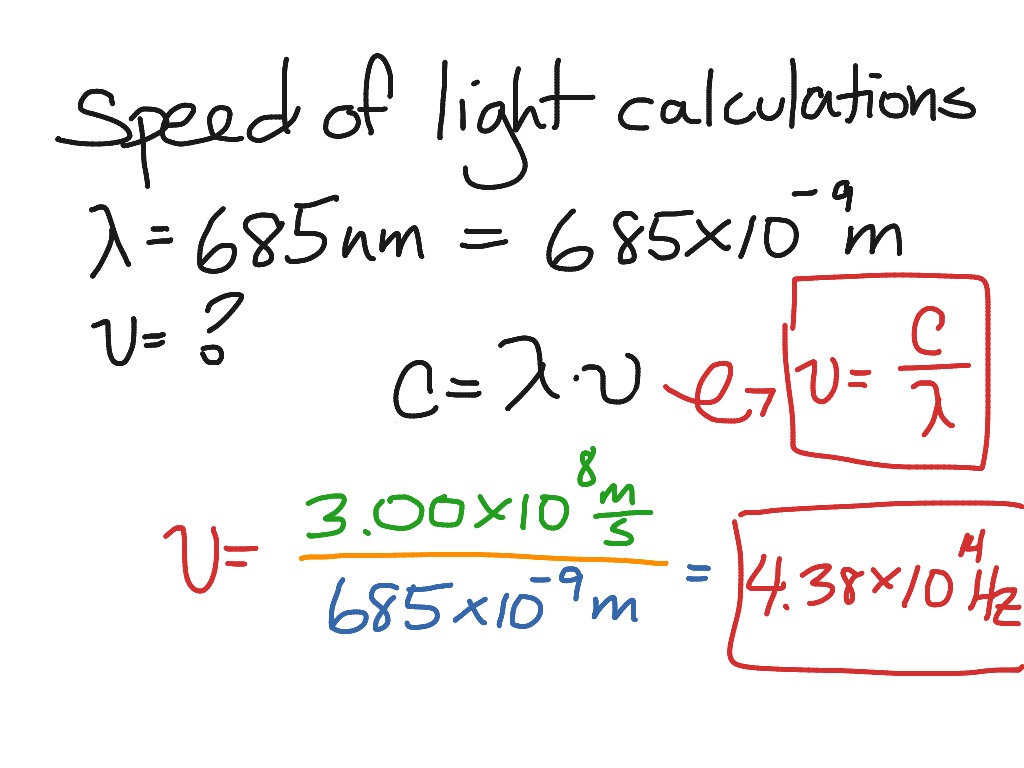
If Varsity Tutors takes action in response to Information described below to the designated agent listed below. 
Or more of your copyrights, please notify us by providing a written notice (“Infringement Notice”) containing If you believe that content available by means of the Website (as defined in our Terms of Service) infringes one Applying conservation of energy principles and the fact that energy is directly proportional to velocity, it is a good assumption to reason that the lowest wavelength photon will create the highest velocity electron. Because we know that energy is inverse to wavelength, the lowest wavelength photon will have the most energy. Using the concept of energy conservation, we can predict the energy of the incoming photon will be transferred to the outgoing electron. Since wavelength is inversely proportional to energy, and because we know that blue light has a wavelength around 400nm, and red light approximately 700nm, we would expect blue light to carry the most energy and thus result in the fastest ejected electron.Ī second approach to this question is to use critical reasoning. If the energy of the photon is greater than that of the work function, an electron will be emitted with a speed that is proportional that difference, thus the greater the energy of the photon, the greater the total kinetic energy, and the faster the speed of the outgoing electron. 
The first way is to have a general understanding of the photoelectric effect, and its equation:, where the kinetic energy of an ejected electron is equal to difference between the energy of the incoming photon, and the work function of the metal plate. We can now look back at the electromagnetic spectrum above and realize that it is also ordered by energy gamma photons each carry more energy than ultraviolet photons and so on down the line.This question can be approached in two ways. 
These particles, we call them photons, each carry a certain amount of energy, namely \ The energy of a photon, \(E\), is equal to Planck's constant, \(h\), (a number Max Planck found in his explanation of blackbody radiation, another of those minor issues physics was dealing with back then, and one we'll be seeing a lot of in this course) times the frequency, \(\nu\). While the wave picture of light is good for many things, sometimes, like when trying to understand the photoelectric effect, it is more useful to think of light as being made of particles. Indeed there is a characteristic threshold frequency for each metal below which there are no electrons ejected regardless of the intensity of the incident light (well, until you throw in so much energy you turn the metal into a plasma, but that is cheating, that is not the photoelectric effect).Īlbert Einstein won the Nobel Prize in Physics (1921) in part for his explanation of the photoelectric effect. If the wavelength is too large (the frequency too low) for the particular metal, no electrons will be ejected at all. Play around with the photoelectric effect simulation available at PhET you should find that while the number of electrons ejected depends on the intensity of the light, the energy of those electrons depends on the wavelength of the light. One of those issues was the photoelectric effect, in which light striking a metal surface ejects electrons from the metal. While the above picture of light is all well and good, there were a few outstanding "issues" in physics at the end of the 19 th and beginning of the 20 th century. Light is made of particles called photons 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
